WHO approves first malaria treatment for newborns, marking major global health breakthrough
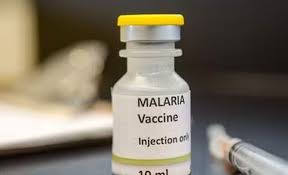
NEW DELHI: In a significant milestone for global healthcare, the World Health Organization has approved the first-ever malaria treatment specifically designed for newborns and young infants, addressing a long-standing gap in pediatric care.
The newly approved treatment is expected to play a crucial role in reducing infant mortality rates, particularly in malaria-endemic regions such as sub-Saharan Africa and parts of Asia, where newborns remain highly vulnerable to severe infections.
For decades, healthcare providers have relied on modified doses of existing malaria drugs for infants, often without formulations tailored to their specific physiological needs. The WHO’s latest approval follows extensive clinical evaluation to ensure the drug meets global standards of safety, efficacy, and quality for neonatal use.
Experts believe this development will strengthen malaria control programmes worldwide and improve survival outcomes among the youngest patients. Malaria continues to be a major public health challenge, causing hundreds of thousands of deaths annually, with children under five accounting for a significant share.
The introduction of a dedicated newborn treatment is being hailed as a critical step toward reducing preventable child deaths and advancing global efforts to combat infectious diseases.